Graphene is a material in which there is only one of these layers of graphite, that is, the fourth carbon bonded electron remaining as a free electron. Graphene has become a unique material due to its excellent electrical conductivity and thermal conductivity, high density and mobility of charge carriers, optical conductivity and mechanical properties. Join us to share useful information about nano graphene nanotech, differenecs graphene and graphite, and more.

Differenecs between graphite & graphene
 Graphene is the name of one of the carbon allotropes. It is a two-dimensional honeycomb crystal structure in which each carbon atom is bonded to three other carbon atoms by means of its three electrons of capacity, with three hybridized SP2 bonds. A residual capacitance electron is also present on the entire graphene plane and between all the atoms shared and the free electron. In graphite (another carbon allotrop), each of the four quaternary carbon atoms, with three covalent bonds, are attached to the other three carbon atoms and form an extensive network. This layer itself is located on a very similar layer, giving the fourth valence electron a chemical bond, but this fourth electron bond is of the van der Waals type, which is a weak bond. For this reason, graphite layers are easily adhered to each other and can be applied to the tip of the pencil.
Graphene is the name of one of the carbon allotropes. It is a two-dimensional honeycomb crystal structure in which each carbon atom is bonded to three other carbon atoms by means of its three electrons of capacity, with three hybridized SP2 bonds. A residual capacitance electron is also present on the entire graphene plane and between all the atoms shared and the free electron. In graphite (another carbon allotrop), each of the four quaternary carbon atoms, with three covalent bonds, are attached to the other three carbon atoms and form an extensive network. This layer itself is located on a very similar layer, giving the fourth valence electron a chemical bond, but this fourth electron bond is of the van der Waals type, which is a weak bond. For this reason, graphite layers are easily adhered to each other and can be applied to the tip of the pencil.
Does graphene come from graphite?
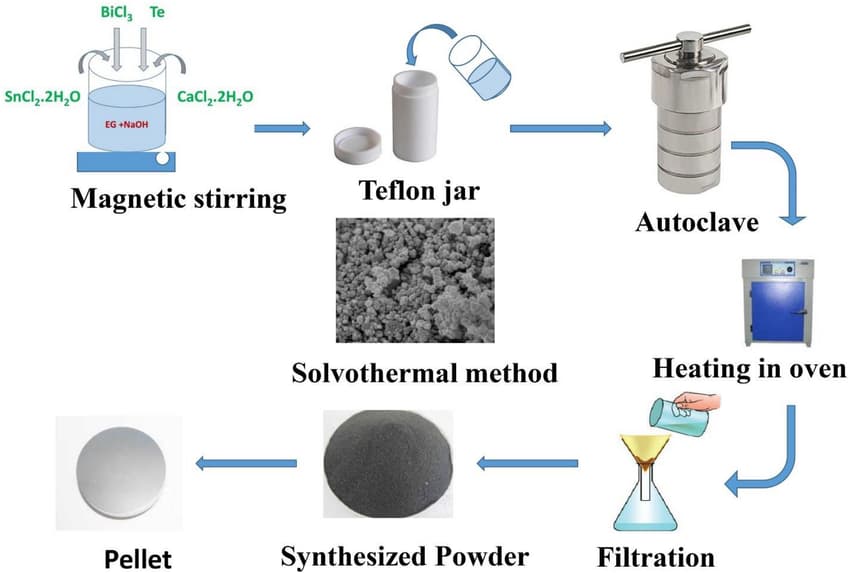
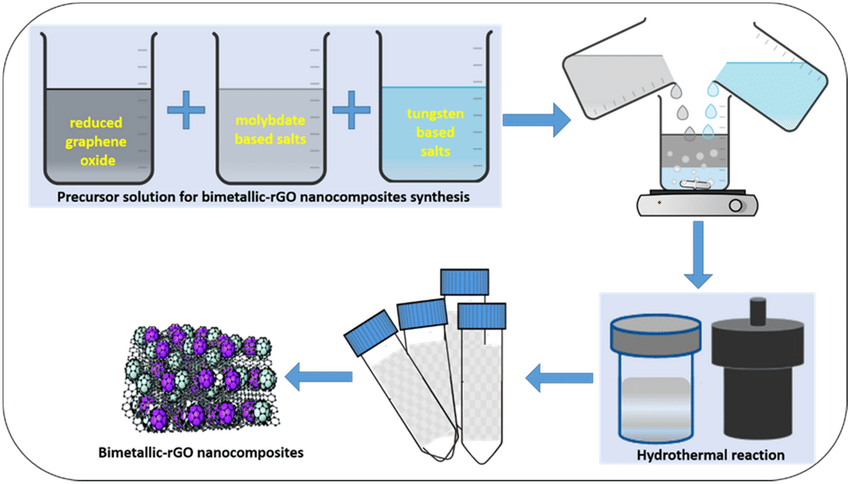
 There are a wide variety of methods used to make graphene today, the most common being mechanical peeling, chemical peeling, synthetic and chemical vapor deposition (CVD). Some other methods, such as carbon nanotube fission and microwave fabrication, have also been used recently. Here is an overview of the methods of graphene fabrication: Bottom-up, Aryan growth, Thermal fission, Plasma, hermal, up to down, Mechanical peeling, Adhesive tape, Atomic Force Microscopy Tips, Chemical peeling, Supercritical Fluids, Chemical synthesis, With ultrasonic waves, Chemical method, Molten salt. Generally, it can be said that graphene is made from graphite. You might be interested to know that monolayer graphene was first produced and reported by Novoselov Group for the first time in years. They used adhesive tape to separate the graphene layers from the substrate surface.
There are a wide variety of methods used to make graphene today, the most common being mechanical peeling, chemical peeling, synthetic and chemical vapor deposition (CVD). Some other methods, such as carbon nanotube fission and microwave fabrication, have also been used recently. Here is an overview of the methods of graphene fabrication: Bottom-up, Aryan growth, Thermal fission, Plasma, hermal, up to down, Mechanical peeling, Adhesive tape, Atomic Force Microscopy Tips, Chemical peeling, Supercritical Fluids, Chemical synthesis, With ultrasonic waves, Chemical method, Molten salt. Generally, it can be said that graphene is made from graphite. You might be interested to know that monolayer graphene was first produced and reported by Novoselov Group for the first time in years. They used adhesive tape to separate the graphene layers from the substrate surface.
Graphene is the future of nanotechnology
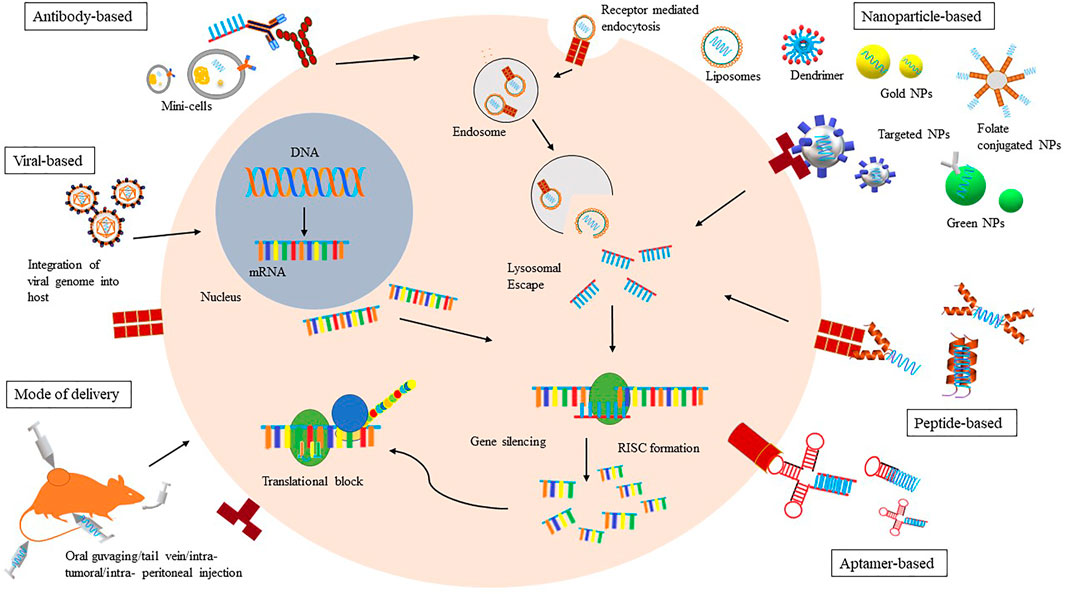
 Graphene has been hailed by many as the “wonder material” of the 21st century. Since graphene is just a single layer of carbon atoms connected in a hexagonal pattern, it is also extremely thin and lightweight, and therefore an attractive material for nanotechnology applications. Graphene nanoparticles has the potential to take off in the field of biomedical research as an ideal material to build medical devices that interact with cells. Graphene, the thinnest known two-dimensional material at only an atom thick, has been in high demand since its discovery due to its strength, flexibility and conductivity.
Graphene has been hailed by many as the “wonder material” of the 21st century. Since graphene is just a single layer of carbon atoms connected in a hexagonal pattern, it is also extremely thin and lightweight, and therefore an attractive material for nanotechnology applications. Graphene nanoparticles has the potential to take off in the field of biomedical research as an ideal material to build medical devices that interact with cells. Graphene, the thinnest known two-dimensional material at only an atom thick, has been in high demand since its discovery due to its strength, flexibility and conductivity.
It can also be added to the graphene properties and uses that a graphene coating is capable of controlling water evaporation by stopping evaporation on hydrophilic surfaces and accelerating evaporation on hydrophobic surfaces. Evaporation of water droplets is a complex phenomenon and plays an important role in nature and industry. Understanding the atomic-scale evaporation mechanism and controlling evaporation is crucial for applications such as heat transfer and body temperature control.
Here’s What You Need to Know About Graphene
 Natural graphene is a metal half or conductive half with a band gap of zero. Understanding the graphene electronic structure is the first step to finding the graphite strip structure. Wallace noticed the linearity of the E-k relation (energy and crystal wave number) near the hexagonal region of the two-dimensional graphene hexagonal Brillouin for low energies, leading to an effective mass of zero for electrons and holes. Because of this low energy dispersive linear relationship, the electrons and holes near these six points, two of which are non-identical, behave like relativistic particles, which are described by the Dirac equation for particles with integer spin.
Natural graphene is a metal half or conductive half with a band gap of zero. Understanding the graphene electronic structure is the first step to finding the graphite strip structure. Wallace noticed the linearity of the E-k relation (energy and crystal wave number) near the hexagonal region of the two-dimensional graphene hexagonal Brillouin for low energies, leading to an effective mass of zero for electrons and holes. Because of this low energy dispersive linear relationship, the electrons and holes near these six points, two of which are non-identical, behave like relativistic particles, which are described by the Dirac equation for particles with integer spin.



Your comment submitted.